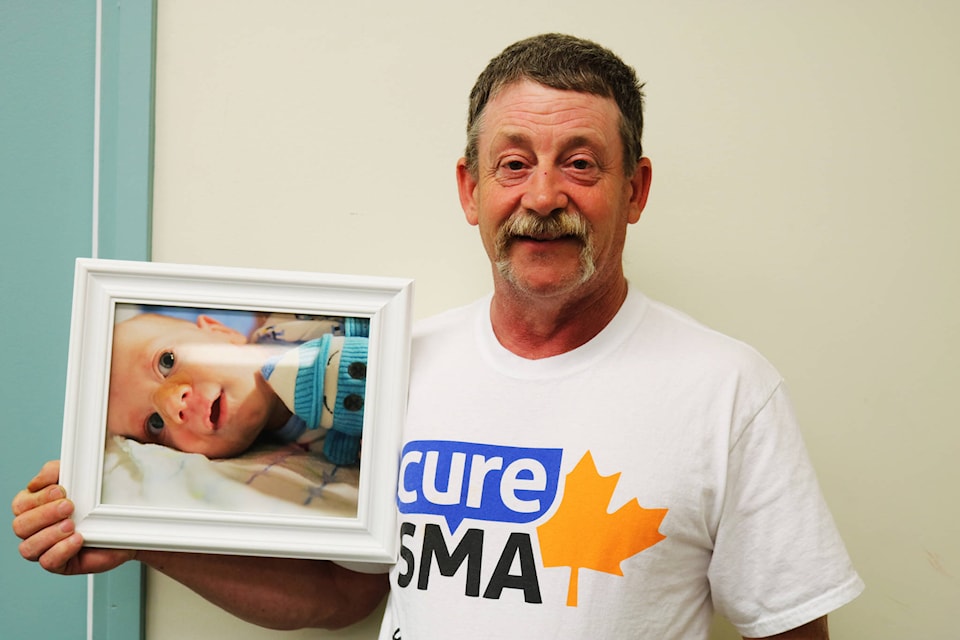
Joe Bramsleven can still hear the clicks the IV machine made as it pumped fentanyl into his dying six-month old son, Kelel.
Bramsleven, along with his wife Tammy, made the decision to take their terminally ill son off of his breathing machine earlier that day.
Kelel had been suffering for a while now and his demise was imminent, so the couple wanted to at least ease his pain. Kelel died in his parents arms within an hour of being taken off his breathing machine; the couple continued to cradle Kelel for six hours after his death. Kelel passed away on Feb. 11, 2015 at Canuck Place Children’s Hospice.
“I’ll never forgive myself in my life,” Bramsleven said. “I hope I rot in hell for what I’ve done.”
Kelel was born on July 31, 2014 with spinal muscular atrophy (SMA) a rare genetic disorder that affects one in 6,000 babies.
According to SMA Europe, the disease is caused by a weak set of nerve cells called lower motor neurones that run from spinal cord to the muscles. These cells carry messages that make movement, like walking, possible.
In order for lower motor neurones to be healthy they need to produce survival motor neuron protein (SMN). The ability to do this is controlled by a gene called survival motorneuron 1 (SMN1). A person born with two faulty SMN1 genes has SMA.
People with two defective SMN1 genes aren’t able to produce the appropriate amount of SMN protein needed for lower motor neurones to be healthy. This causes the lower motor neurones to deteriorate making it difficult to send messages to the muscles.
This severely limits movement and eventually the lack of movement causes the muscle to waste away and this is known as muscular atrophy. People with SMA have a limited ability to sit up, eat or sometimes even breathe.
There are four types of SMA with type one being the most severe — the life expectancy for babies with type one is less than 18 months — to least severe which is type four. Kelel, a type one, had one of the worst cases SMA in Canadian history, Bramsleven said.
Kelel was paralyzed from the chin down at birth. He couldn’t swallow or cough and eventually could barely move. Bramsleven said a simple mistake like picking him up wrong could kill him. One nurse couldn’t handle the pressure and quit on the couple.
“It’s 24-hour care, 365 days a year including Christmas,” Bramsleven said. “Everything about what you wanted to do in life — your aspirations, your desires, your dreams, your goals — everything is finished.”
Bramsleven and Tammy attended Cure SMA Canada’s annual summer camp for Western families effected by SMA in Hope last month. Kids did typical summer camp activities like crafts, kayaking and campfires that have been modified for various levels SMA.
Since the death of their son, Bramsleven and Tammy have been advocates for SMA and were invited by Cure SMA Canada executive director Susi Vander Wyk to share their story with other parents of SMA children at the camp.
Bramsleven told parents about saving his son’s life by giving him mouth-to-mouth resuscitation at his christening; friends that avoided him during his son’s health issues; and how his business Dynamic Cleaning Services suffered as he lost multiple contracts, which he still hasn’t recovered from. But halfway through the tale one woman asked him to stop.
“It hurt but [I] understood,” Bramsleven said. “At that point [I] felt alienated.”
The parents at the camp had either just begun or were halfway through their journey with SMA and viewed the couple as the bleak future that awaited them, Bramsleven said.
Parents also heard the latest information regarding Cure SMA Canada’s fight for access to Spinraza, a drug used to treat SMA created by American biotechnology company Biogen.
Spinraza increases SMN protein by targeting a gene called survival motor neuron 2 (SMN2), which like SMN1 produces the SMN protein. Without Spinraza, SMN2 cannot produce enough SMN protein to keep lower motor neurons healthy.
Although Spinraza does not cure SMA completely, it does allow those afflicted with the disease to live.
According to head of market access for Biogen Canada Farah Jivraj, SMA patients that have taken the drug have shown vast improvements in mobility.
Some people that couldn’t eat gained the ability to do so; others who struggled to walk to end of their driveway were able to walk much further distances, Jivraj said.
The life-saving drug comes at a cost — $750,000 for the first year. Treatment goes down to $375,000 for every year after that. SMA patients will need to take the drug for their lives.
Spinraza is expensive because of the high cost of research and development that went into creating the drug, Jivraj said.
Biogen conducted 10 clinical trials — which is unprecedented for a rare disease — that had over 300 patients, Jivraj said.
In 2017 Health Canada authorized the sale of Spinraza in Canada.
In order for a drug to be considered for a provincial drug plan it needs a recommendation from the Canadian Agency for Drugs and Technologies in Health (CADTH).
CADTH makes reimbursement recommendations for federal, provincial, and territorial public drug plans after analyzing the cost-benefits of medications. CADTH recommended the government only cover Spinraza for type one SMA patients that were diagnosed before they turned seven months old.
Kelel was diagnosed with SMA just as Biogen was beginning its clinical trials for Spinraza. Bramsleven and Tammy tried to get their son on the trials but were denied. An attempt by Cure SMA Canada to get him special access to the drug also failed.
Even during trials the drug showed significant results. Biogen had to stop the trials early and convert to active treatment because it was deemed unethical to continue to provide some patients with placebos when the drug was working so well, Jivraj said.
“It is possible that Kelel could be still alive had he been able to access Spinraza,” Vander Wyk said.”We don’t know that for sure but other children did very well on it.”
A total of 41 countries including Australia, Belgium, and the Netherlands, provide government reimbursement for Spinraza, Jivraj said.
Biogen resubmitted an application requesting reimbursement for SMA type one, two and three patients to CADTH.
“We continue to work with the government to find a path forward to be able to provide or gain access to Spinraza for Canadian patients,” Jivraj said.
Jivraj said some private insurers cover Spinraza but did not name any specific company.
CADTH was unable to comment on Spinraza, its past recommendation or Biogen’s current application because of the ongoing review.
“The ultimatum in this is only one thing — death,” Bramsleven said. “It’s a life saving drug: you either get it or you die. It’s that simple.”
Bramsleven and Tammy are in the process of creating SMA Battleground, a foundation that supports the day-to-day costs of having a child with SMA like getting new wheelchairs and food.
“I swore to my son as he was passing in my arms that I will do everything in my power ‘till the day I die to either find a cure or to help other families with SMA,” Bramsleven said.
Bramsleven encourages anyone dealing with or knows someone that is dealing with SMA in northern B.C. to give him a call at 250-847-0756.
Anyone interested in learning more about SMA or donating to fund research for SMA can do so by visiting CureSMA.ca.